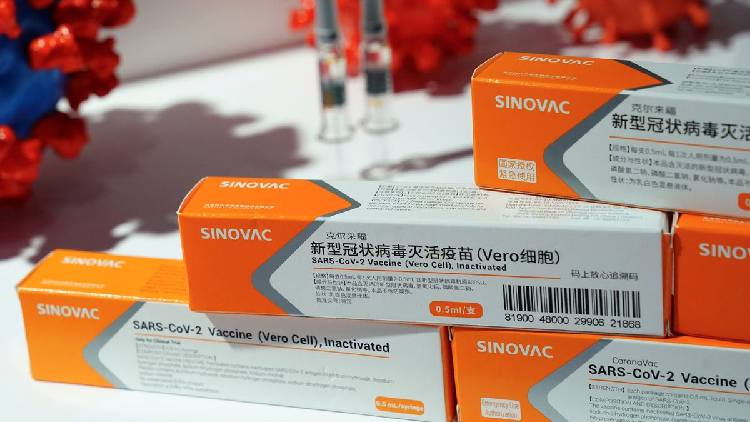
Sinovac Vaccine
Sinopharms other vaccine developed via a Wuhan unit and not listed for emergency use by the WHO has an efficacy rate of around 73. Sinovac Biotech Ltds vaccine was less potent than Pfizer Incs at stopping Covid-19 in Chile where the two shots were used simultaneously allowing the first real-world comparison of the.

China Approves First Mixed Vaccine Trial As Delta Spreads France 24
There 55 of the population is fully vaccinated and among that cohort almost 80 received Sinovac.
Sinovac vaccine. After 90 days of the second Sinovac jab people can receive the mRNA shot from Pfizer. SINGAPORE From next Tuesday 10 August those in Singapore who receive COVID-19 jabs that are not under the national programme but are under the World Health Organisations WHO. The five most common adverse events of Sinovacs COVID-19 vaccine are elevated blood pressure headache injection site pain dizziness and rash while the top five side effects of AstraZenecas.
The SinoVac vaccines efficacy data. WHOs Strategic Advisory Group of Experts on Immunization. Turkey started to administer Sinovac vaccines in mid-January and at least eight million people have been injected with the vaccine accounting for more than 10 percent of the population.
However this vaccine is an inactivated vaccine with an adjuvant that is commonly used in many other vaccines with a well-documented safety profile such as Hepatitis B and. The Sinovac vaccine contains an inactivated form of coronavirus that cannot cause the disease. The Beijing-based biopharmaceutical company Sinovac is behind the CoronaVac an inactivated vaccine.
The Philippines FDA said more time is needed to determine. 6 August 2021 442 am 2-min read. CoronaVac is a COVID-19 vaccine produced by Sinovac Biotech a China-based pharmaceutical company with headquarters in Beijing.
The Sinovac-CoronaVac product is an inactivated vaccine. Sinovacs vaccine became the worlds most produced vaccine at the end of May according to Airfinity analysis surpassing PfizerBioNTech. The country has administered more than 18 million Sinovac vaccine doses.
Sinovacs commercialized vaccines include CoronaVac COVID-19 vaccine Inlive Enterovirus 71 vaccine Anflu influenza vaccine Healive hepatitis. Those given Sinovac Sinopharm or AstraZeneca jabs counted as fully vaccinated from 10 Aug. A recent study published in the New England Journal of Medicine which involved 102 million participants in Chile showed that the Sinovac vaccine was.
It also has a substance that helps strengthen the immune response to the vaccine. The Sinovac CoronaVac vaccine also uses a virus to create immunity. The government will offer a third shot to increase the populations immunity to Covid-19 variants such as.
Malaysia has given conditional approval for a COVID-19 vaccine made by Chinas Sinovac to be used on young people aged between 12 and 17 its health ministry said on Friday. It works by using killed viral particles to. But according to the Ministry of Health 73 of cases in the intensive care unit between June.
The available data on the Sinovac-CoronaVac COVID-19 vaccine in pregnant women are insufficient to assess either vaccine efficacy or possible vaccine-associated risks in pregnancy. However instead of adenovirus the vaccine uses inactivated particles of the SARS-CoV-2. SINGAPORE There is a significant risk of vaccine breakthrough with the Sinovac vaccine or CoronaVac with international evidence showing that many who had taken it were later infected with COVID-19 said the Ministry of Healths MOH director of medical services Kenneth Mak on Friday 18 June.
In a statement the health ministry said Malaysias drugs regulator advised priority for Sinovacs product among teenagers without co-morbidities or allergy problems or otherwise deemed. But their case numbers also rebounded in late February with 37303 new cases recorded on March 30 the highest single-day figure since the outbreak on March 11 last year. Its easy storage requirements make it very manageable and particularly suitable for low-resource settings.
Covid What Do We Know About China S Coronavirus Vaccines Bbc News
Coronavirus Digest China S Sinovac Says Vaccine Stops Covid 19 Deaths News Dw 06 02 2021

Sinovac Vaccine Production Line In Egypt To Start Operating Soon Global Times
Coronavirus Digest China S Sinovac Vaccine 78 Effective Says Brazil News Dw 07 01 2021
Another Covid 19 Vaccine To Boost Rollout
Sinovac Vaccine Has 97 Seroconversion Rate In Indonesia Trial Bloomberg

China S Sinovac Vaccine S Low Efficacy Due To High Risk Group Ceo Bloomberg
China To Only Allow Foreign Visitors Who Have Had Chinese Made Vaccine China The Guardian
Sinovac Covid 19 Vaccine Safe For Ages 3 To 17 In Small Early Trial Devex
Paul Ehrlich Institut News Start Of The Rolling Review Procedure For Sinovac S Whole Virus Vaccine Called Covid 19 Vero Cell Inactivated At The European Medicines Agency

Sinovac Reports Positive Data From Phase I Ii Trials Of Coronavac
Singapore Excludes Sinovac Shots From Covid 19 Vaccination Tally Nikkei Asia
Gavi Signs Agreements With Sinopharm And Sinovac For Immediate Supply To Covax Gavi The Vaccine Alliance

Eu Regulator Examines Sinovac S Covid 19 Vaccine

Third Shot Of Sinovac Covid 19 Vaccine Offers Big Increase In Antibody Levels Study Global Times

Coronavirus Who Approves Sinovac Covid Vaccine For Emergency Use News Dw 01 06 2021
Who Grants Emergency Authorisation To Sinovac Vaccine Latest Updates
.JPG)